Why PMA-Zeolite?
PANACEO’s patented micro-activation makes the difference: maximum binding of harmful substances, clinically proven, CE-certified — Made in Austria.
Discover PMA-Zeolite products
What is PMA-Zeolite?
PMA-Zeolite is a patented medical device by PANACEO based on clinoptilolite zeolite of volcanic origin. Through the company’s proprietary PMA activation process (Patented Micro Activation), the crystal structure of the mineral is optimized and its natural adsorption power for harmful substances is enhanced many times over.
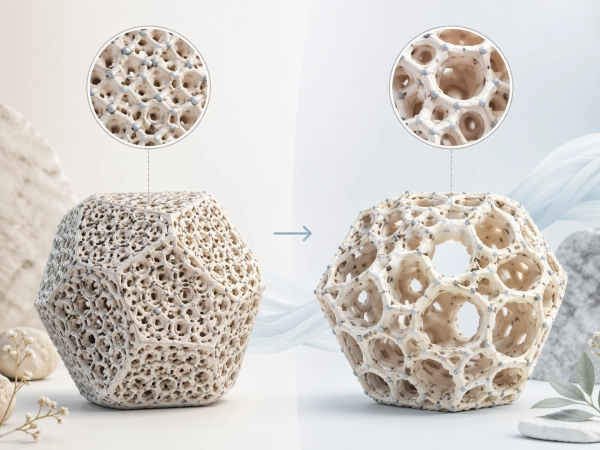
Not all zeolite is the same. Quality, purity, and effectiveness can vary considerably depending on origin, processing, and particle size. With PMA technology, PANACEO has developed a process that specifically enhances the natural properties of clinoptilolite zeolite.
The result: PMA-Zeolite has an increased inner surface area, an optimized crystal structure, and a uniform particle size of 1–10 micrometers — the optimal range for maximum binding of harmful substances without nanoparticles.
All PANACEO medical devices are based on this PMA-activated zeolite.
PMA — Patented Micro Activation
A three-stage patented process that transforms zeolite from a raw mineral into a high-performance medical device.
Raw material selection
Only the purest clinoptilolite of volcanic origin is selected. Strict incoming inspections verify purity, clinoptilolite content, and freedom from harmful substances.
Mechanical activation
The patented PMA process mechanically optimizes the crystal structure — without chemicals, without heat. The inner surface area is increased and the pores are opened.
Quality control
Each batch is tested for particle size (1–10 µm), adsorption capacity, and purity. Only after passing inspection is it approved for products.
PMA-Zeolite vs. regular zeolite
What happens when zeolite is not activated? The differences are clinically measurable.
PMA-Zeolite (PANACEO)
- ✓ Patented micro-activation
- ✓ Optimized crystal structure
- ✓ Increased inner surface area
- ✓ Particle size 1–10 µm (no nano)
- ✓ CE-certified Class IIb
- ✓ Clinically tested effectiveness
- ✓ ISO 13485 quality standard
- ✓ Made in Austria
Unactivated zeolite
- ✕ No activation
- ✕ Natural, irregular structure
- ✕ Smaller surface area
- ✕ Uneven particle size
- ✕ Often without CE certification
- ✕ Rarely clinically tested
- ✕ Different quality standards
- ✕ Different countries of origin
Why you should pay attention to certification
PMA-Zeolite is certified as a IIb medical device. This means: strict testing, proven effectiveness, documented safety.
CE certification Class IIb
CE class IIb for ingestible medical devices and suitable for long-term use. Stricter requirements than for dietary supplements — comparable to pharmaceutical standards.
ISO 13485 quality management
International standard for the manufacturing of medical devices. Every step — from raw material selection to packaging — is documented and controlled.
Clinically tested safety
Studies confirm: no aluminum is released, even after months of use. The high silicon content ensures the stability of the crystal lattice.
100% Made in Austria
Raw material selection, PMA activation, and final inspection — everything takes place in Carinthia, Austria. Short distances, highest standards, full traceability.
Clinical studies on PMA-Zeolite
The effectiveness and safety of PMA-Zeolite are documented in numerous peer-reviewed studies.
Strengthening the intestinal barrier
Double-blind, placebo-controlled study: PMA-Zeolite improves intestinal wall integrity and significantly reduces inflammatory markers. Zonulin as a leaky gut marker was clearly reduced.
Selective binding of heavy metals
PMA-Zeolite selectively binds lead, cadmium, and arsenic in the gastrointestinal tract — without negatively affecting mineral balance. Controlled clinical study.
No aluminum release
After 3 months of daily use: aluminum levels in the blood remained unchanged. The stable SiO₄/AlO₄ lattice releases no aluminum — clinically confirmed.
Reduction of oxidative stress
PMA-Zeolite shows antioxidant properties: reduction of malondialdehyde (MDA) and other oxidation markers with regular intake.
The right PMA-Zeolite product for you
All PANACEO medical devices are based on PMA-Zeolite — CE-certified, clinically tested, made in Austria.